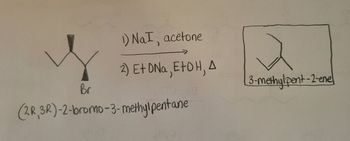From each of the following pairs select the compound that will react faster with sodium iodide in acetonea 2 Chloropropane or 2 bromopropane I IIb 1 Bromobutane or 2 bromobutane I II

a) Give the product of debromination with NaI in acetone solution of the following. (i) Erythro or meso -2,3- dibromobutane (ii) Threo or `(+-)-2,3-` - Sarthaks eConnect | Largest Online Education Community
NAI-150A-1 - 1,4-Naphthoquinone (cas 130-15-4) Solution, 100 µg/mL, in acetone - Ultra Scientific Italia
Here, $\\text{1-4 dichlorohexane (1 mole) + NaI (1 mole)}\\xrightarrow{ Acetone}$ Product of the reaction is:(a) \n \n \n \n \n (b)\n \n \n \n \n (c) \n \n \n \n \n (d)\n \n \
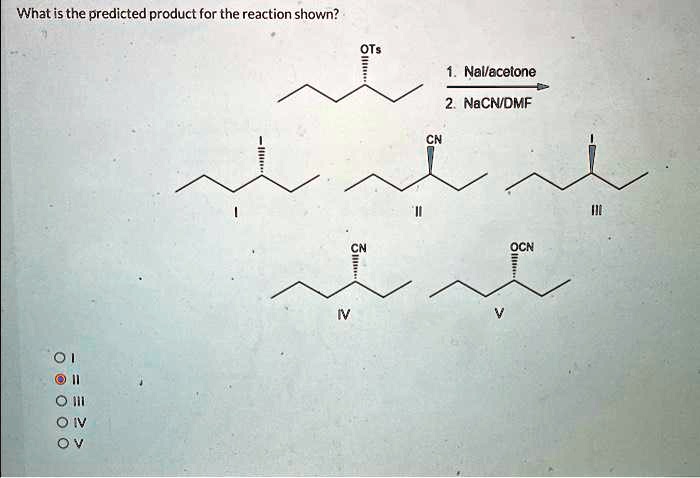
SOLVED: What is the predicted product for the reaction shown? 1. NaI/acetone 2. NaCN/DMF CN OCN Oâ‚‚

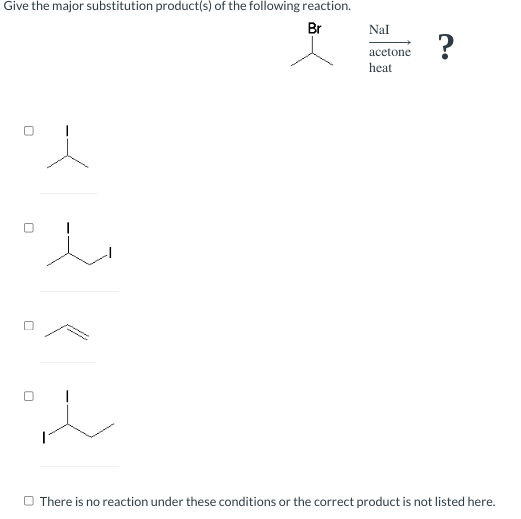
SOLVED: What is the major product for the following reaction? 1. NaI, acetone 2. NaOH, EtOH, heat CI B) E)

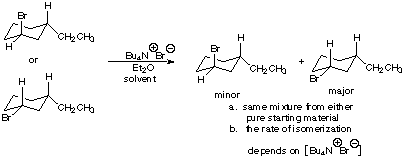
te Nai Acetone 1 → Product Acetone > Product. * Product and reactant related as (1) Identical molecules 21 Diastereomers (3) Enantiomers (4) Two different molecules
Scheme 1 Synthesis of N 6-benzoyl-AdoHyc. (a) NaI-acetone, 69%. (b)... | Download Scientific Diagram