Separation of methyl acetate + methanol azeotropic mixture using ionic liquids as entrainers - ScienceDirect
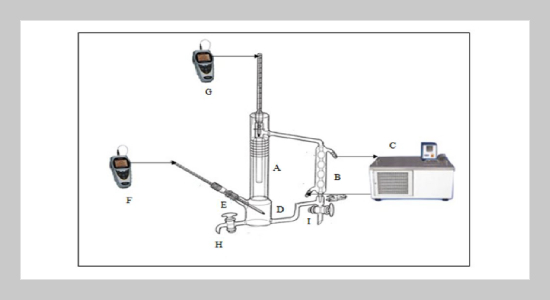
Separation of Azeotrope Mixture Composed of Acetone and Methanol using Various Entrainers - Journal of Applied Science and Engineering

The Design and Optimization of Extractive Distillation for Separating the Acetone/n-Heptane Binary Azeotrope Mixture | ACS Omega

Separation of the Azeotropic Mixture Methanol and Toluene Using Extractive Distillation: Entrainer Determination, Vapor–Liquid Equilibrium Measurement, and Modeling | ACS Omega
Separation of Mixture Containing Maximum Boiling Azeotrope with Extractive Heterogeneous-Azeotropic Distillation

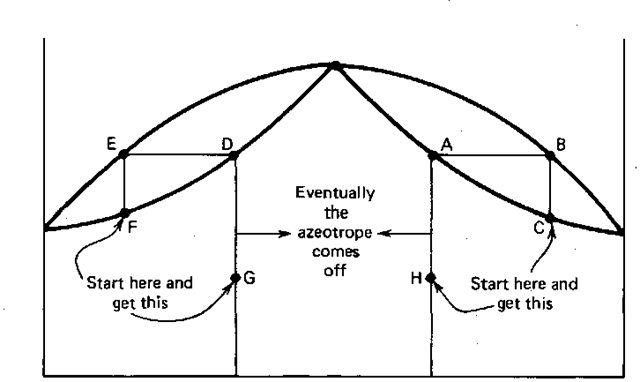
Breaking the Azeotrope between Methanol and Acetone with an Entrainer - Wolfram Demonstrations Project

Separation of acetonitrile/methanol/benzene ternary azeotrope via triple column pressure-swing distillation - ScienceDirect

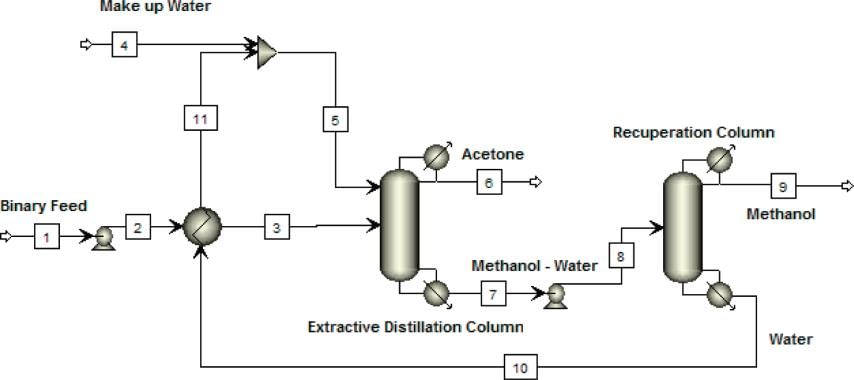
Extractive Distillation of Acetone/Methanol Mixture Using Water as Entrainer | Industrial & Engineering Chemistry Research
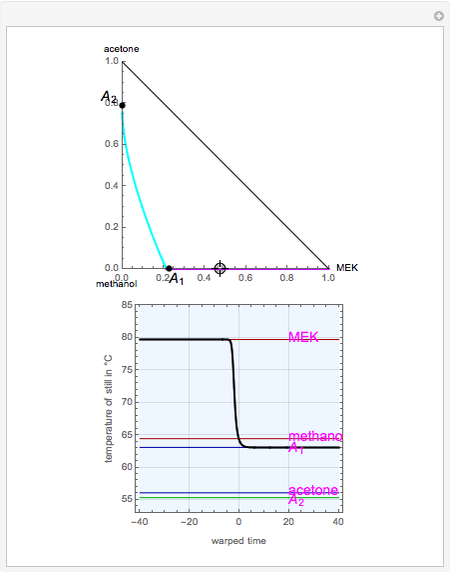
Breaking the Azeotrope between Methanol and Acetone with an Entrainer - Wolfram Demonstrations Project
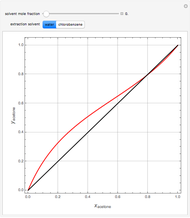
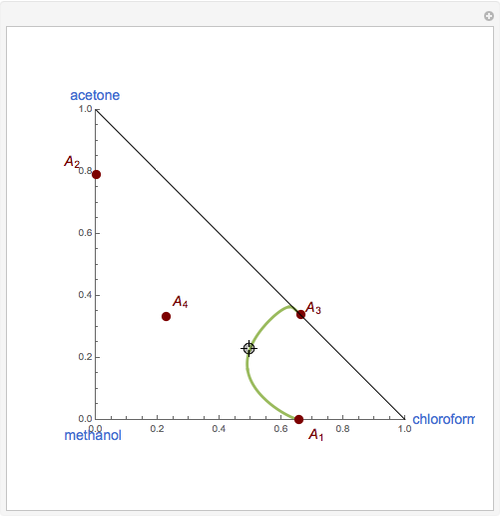
Breaking the Acetone-Methanol Azeotrope with Different Extraction Solvents - Wolfram Demonstrations Project

Separation of the Azeotropic Mixture Methanol and Toluene Using Extractive Distillation: Entrainer Determination, Vapor–Liquid Equilibrium Measurement, and Modeling | ACS Omega

Schematic diagram of the conventional extractive distillation, acetone | Download Scientific Diagram

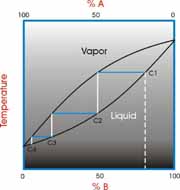
SOLVED: a) For a binary mixture consisting of acetone (1) and methanol (2), it is found that the mixture exhibits an azeotrope at a certain pressure P and temperature T. Ideal gas