The reaction between benzaldehyde derivatives with acetone in presence... | Download Scientific Diagram

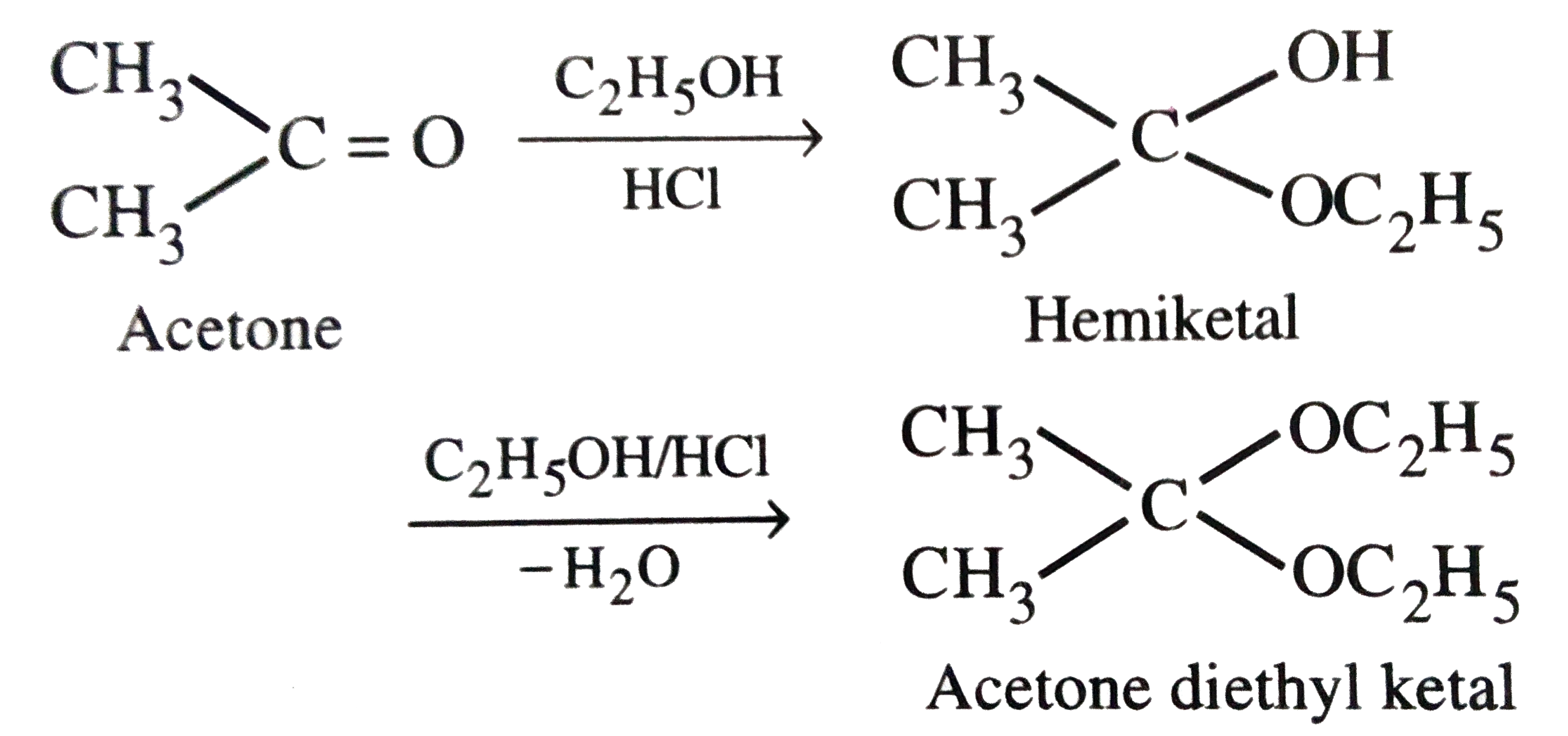
SCHEME 1. (i) a, acetone, 4-MU, Na4-MU; b, K 2 CO 3 , Et 2 O; c, HCl;... | Download Scientific Diagram
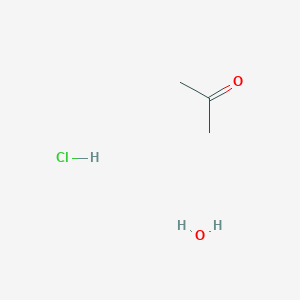
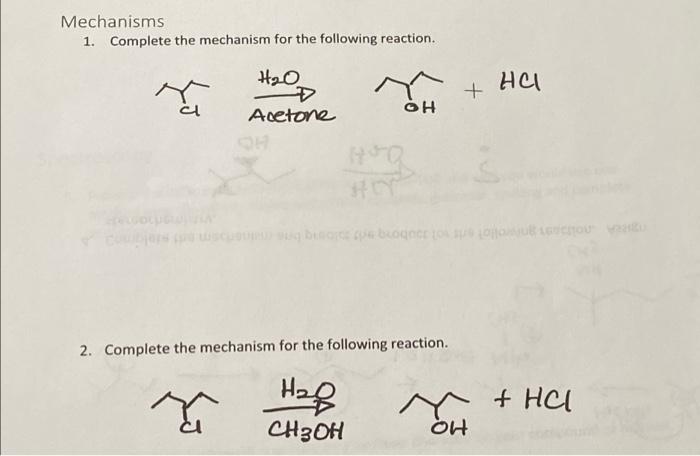
Acetone is treated with excess of ethanol in the presence of hydrochloric acid. The product obtained is - Sarthaks eConnect | Largest Online Education Community
What explosive is formed if I mix acetone, hydrochloric acid and hydrogen peroxide? How do I detonate it? - Quora
Acetone is treated with excess of ethanol in the presence of hydrochloric acid. The product obtained is - Sarthaks eConnect | Largest Online Education Community

Why does acetone remove the dark blue colour of the solution containing iodine, hcl, and starch, when thoroughly mixed? | Homework.Study.com
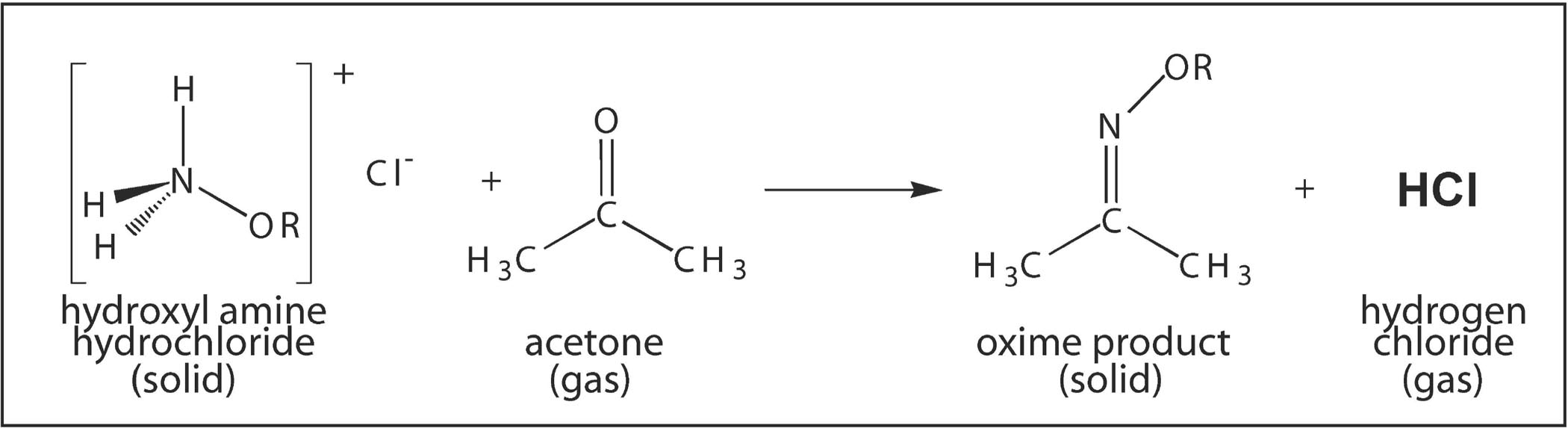
This polymer (B) is obtained when acetone is saturated with hydrogen chloride gas, - Sarthaks eConnect | Largest Online Education Community

Acetone is treated with excess of ethanol in the presence of hydrochloric acid.The product obtained - YouTube

Large Scale Deprotection of a tert-Butoxycarbonyl (Boc) Group Using Aqueous HCl and Acetone | Organic Process Research & Development